Europe
Americas
Asia Pacific
Eurasia & Middle East
Europe
Americas
Asia Pacific
Eurasia & Middle East







EMFACE is the market-first device that simultaneously stimulates facial muscles and remodels skin using a patented combination of HIFES and Synchronized RF technologies.
Designed for clinics looking to deliver natural-looking, non-invasive facial rejuvenation results, EMFACE targets the full facial structure, from muscle to skin.


EMFACE firms the facial framework and lifts the skin using our 2-in-1 technology:
This dual-technology approach allows providers to address facial volume loss, sagging, and visible signs of aging without injectables or surgery.
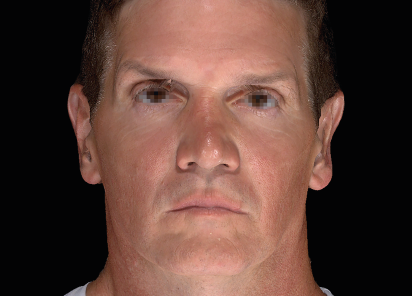
EMFACE delivers measurable lifting and contouring by targeting the muscles and skin that support facial structure.
Clinical results show visible improvements across multiple facial areas.
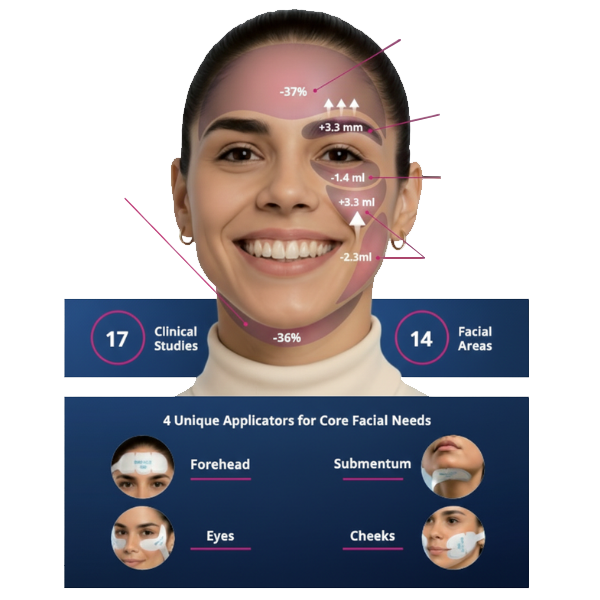
Click on the desired area for EMFACE results.
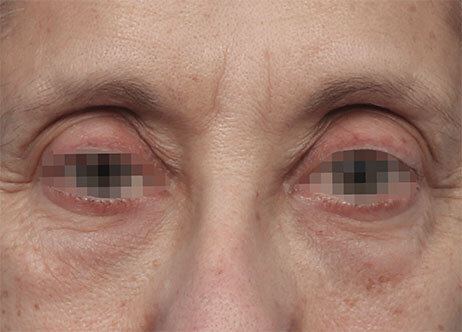
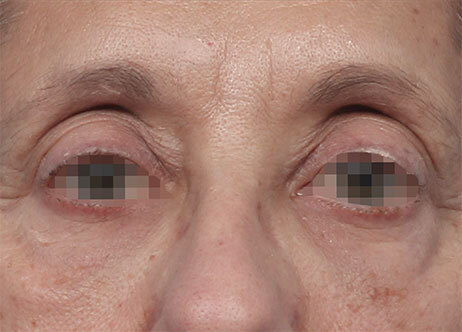
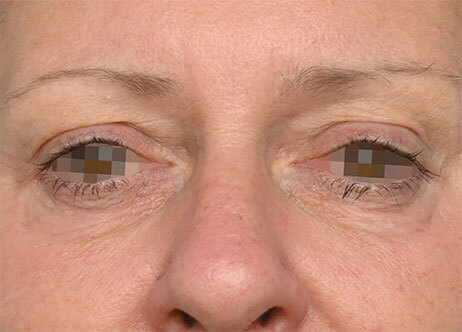
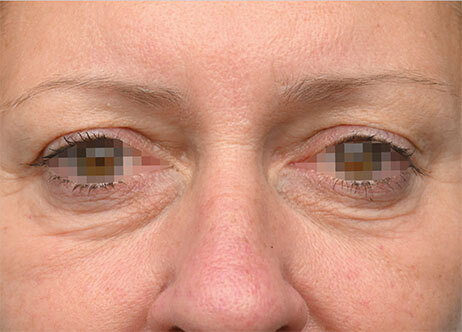
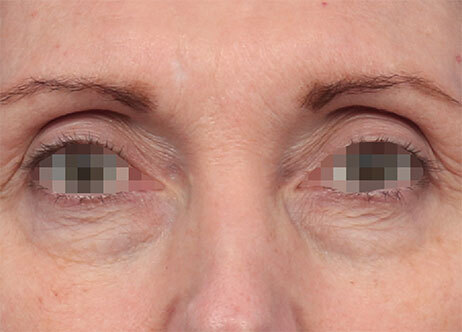
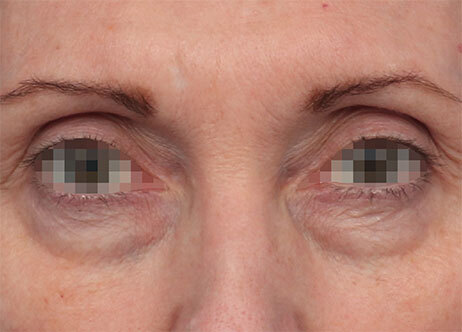
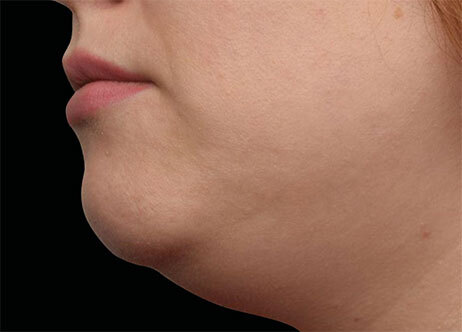
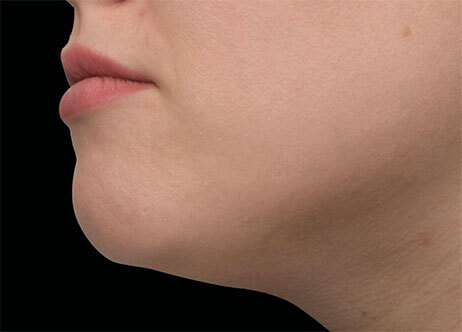
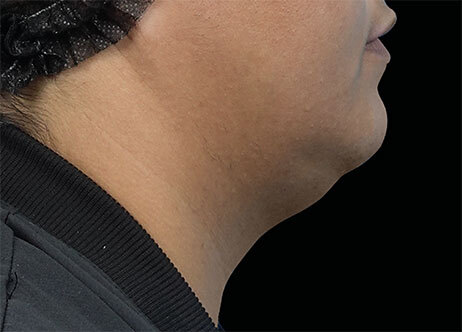
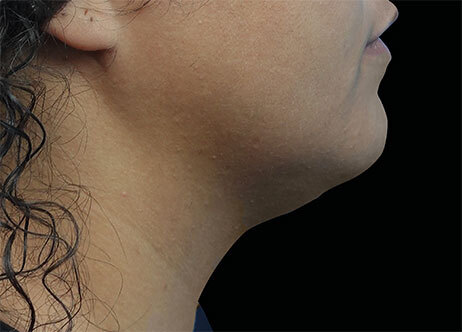
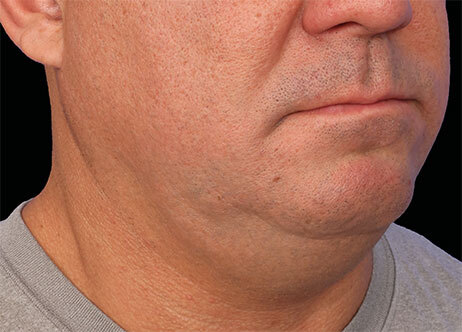
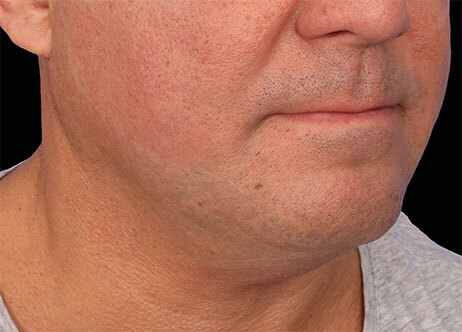
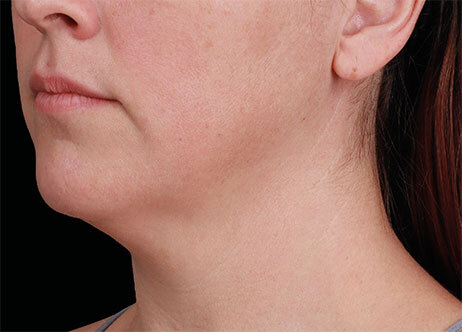
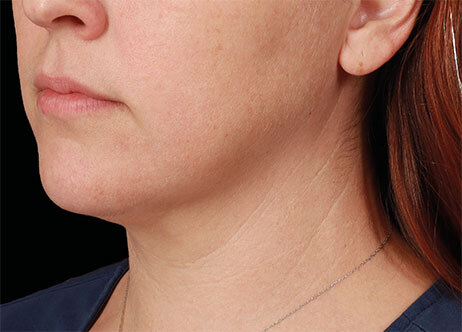


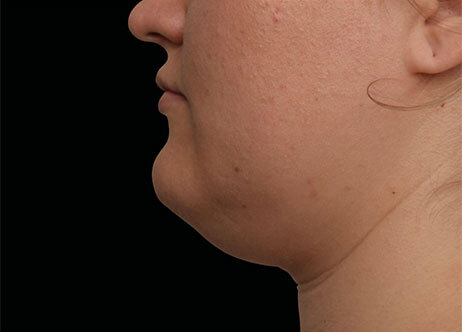
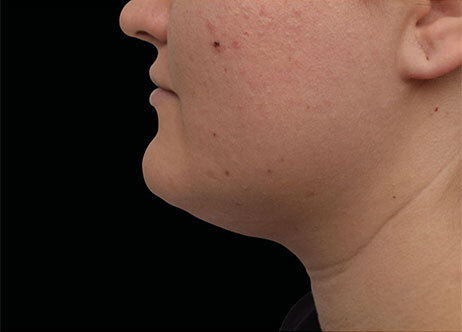
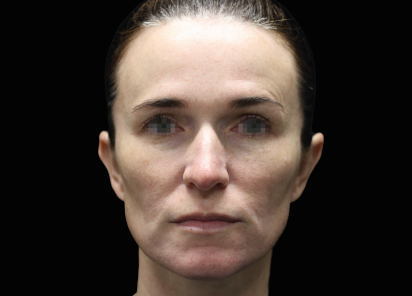
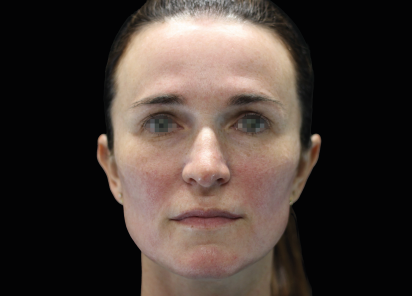
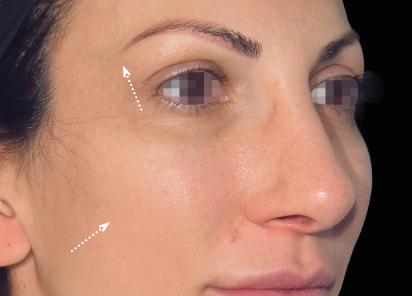
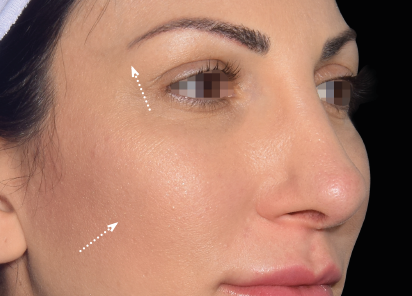


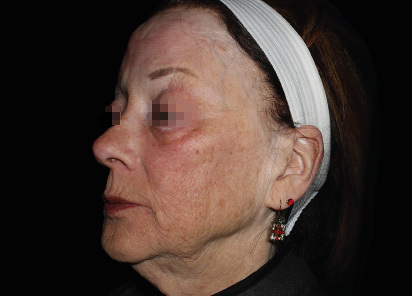
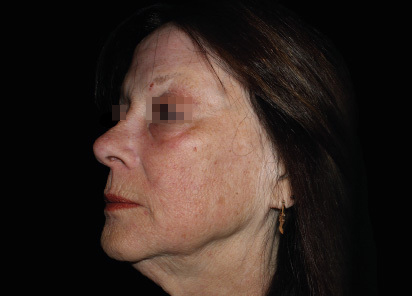

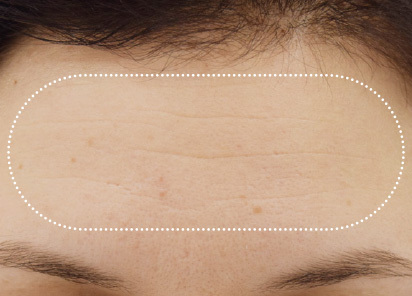
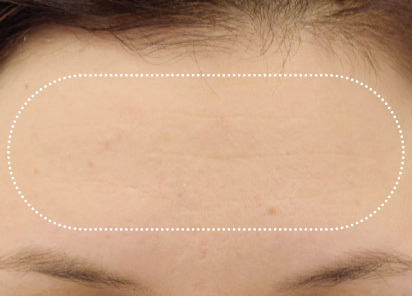
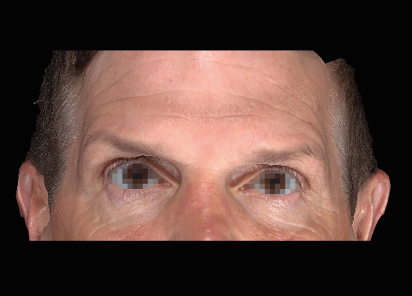
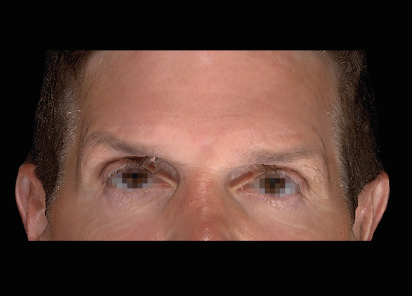
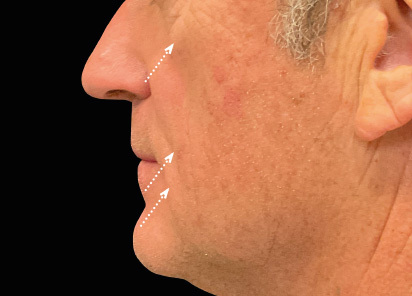
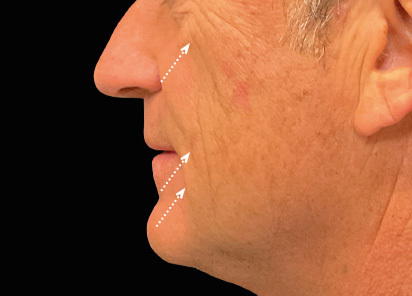
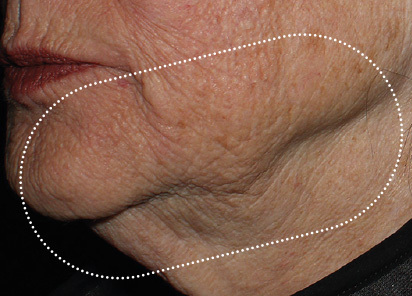
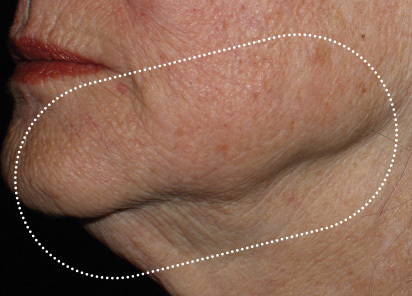
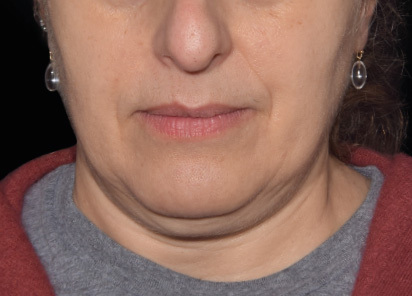
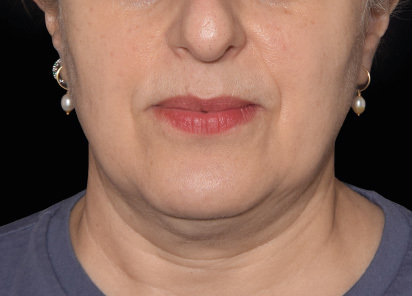
Results and client experience may vary.
EMFACE is ideal for aesthetic clinics looking to offer next-generation facial rejuvenation treatments that support both volume and lift, without the need for injectables.
EMFACE is part of the BTL portfolio, trusted by leading clinics around the world for over 30 years.
BTL supports practices with technologies that are:
Alongside EMFACE, BTL offers a suite of non-invasive systems to help clinics diversify their service offering:
rTMS brain stimulation for mental wellness
Muscle building and fat reduction
Each technology is engineered for measurable results, efficient workflows, and commercial success.
Our team works closely with clinics to ensure seamless implementation and business results.
This product is not available for purchase by the general public.
The findings showed consistent outcomes across the clinical studies.
Patient results and experience may vary.

Become a BTL Partner today.
BTL partners with clinics committed to clinical excellence and innovation.
Evidence based technologies with global clinical adoption
Comprehensive training and marketing support
Dedicated technical service and ongoing partnership
Devices registered and compliant in Australia and New Zealand
Fill in your details and our team will be in touch to discuss your goals and product suitability.
By filling the form I am aware that BTL contacts me to keep me informed about news of BTL Group and about its products and services.
Address:
Administration & Service:
Unit 5 / 10 Enterprise Close
West Gosford NSW 2250
Showroom:
Suite 2.05, Level 2, Building D, Talavera Corporate Center,
12-24 RTalavera Road, Macquarie Park NSW 2113
Email: info@btlmed.com.au
Phone: 0422 608 123
Address:
130 St Georges Bay Rd, Parnell,
Auckland 1052
Email: ehab@btlmed.nz / info@btlmed.nz
Ehab Jirgis , Business Development Manager +64 21 284 2628
Clinical references:
1) Gentile R, Halaas Y. Facial Plast Surg Aesthet Med. 2024;26(1):1-6. doi:10.1089/fpsam.2023.0015,
(2) Frank K, Kaye KO, Casabona G, et al. Aesthet Surg J. 2025;45(5):525-30. doi:10.1093/asj/sjaf009,
(3) Palm, M.; Kilmer, S.M. & Gentile,R. (2025): Novel HIFESTM and Synchronized Radiofrequency Effect on Periorbital Area; presented at 44th ASLMS Annual Conference on Energy-Based Medicine & Science, Orlando, 25 April 2025,
(4) Cohen JL, et al. Dermatol Surg. 2025;51(7):690-5. doi:10.1097/DSS.0000000000004613,
(5) DiBernardo B, Jacob C, Clark-Loeser L. Aesthet Surg J. 2025;45(7):709-14. doi:10.1093/asj/sjaf048.